How and why liquefied gas: production technology and scope of use of liquefied gas
Technologies related to the extraction, transportation and processing of natural gas are developing rapidly. And many today have heard the abbreviations LNG (LPG) and LPG (LNG). Almost every other day, natural gas fuel is mentioned in the news in one context or another.
But, you must admit, in order to have a clear understanding of what is happening, it is important to initially understand how gas is liquefied, why this is done and what benefits it gives or does not give. And there are many nuances in this matter.
To liquefy gaseous hydrocarbons, large high-tech plants are being built. Next, we will carefully understand: why all this is needed and how it happens.
The content of the article:
Why liquefy natural gas?
Blue fuel is extracted from the bowels of the earth as a mixture of methane, ethane, propane, butane, helium, nitrogen, hydrogen sulfide and other gases, as well as their various derivatives.
Some of them are used in the chemical industry, and some are burned in boilers or turbines to generate heat and electricity. Plus, some of the extracted is used as a gas engine fuel.

The main reason for liquefying natural gas is the simplification of its transportation over long distances. If the consumer and the gas fuel well are located on land close to each other, it is easier and more profitable to lay a pipe between them. But in some cases, building a highway is too expensive and problematic due to geographical nuances. Therefore, they resort to various technologies for producing LNG or LPG in liquid form.
Economics and Transportation Security
After the gas is liquefied, it is already pumped in the form of liquid into special containers for transportation by sea, river, road and / or rail.In this case, technologically, liquefaction is a fairly costly process from an energy point of view.
At different plants, this takes up to 25% of the initial fuel volume. That is, to generate the energy needed by technology, you have to burn up to 1 ton of LNG for every three tons of it in finished form. But natural gas is now in great demand, everything pays off.
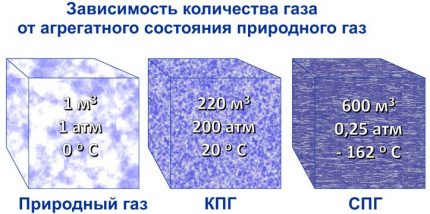
While natural gas is in a liquid state, it is non-combustible and non-explosive. Only after evaporation during regasification, obtained gas mixture turns out to be suitable for burning in boilersand cooking hobs. Therefore, if LNG or LPG is used as hydrocarbon fuel, then they must be regasified.
Use in various fields
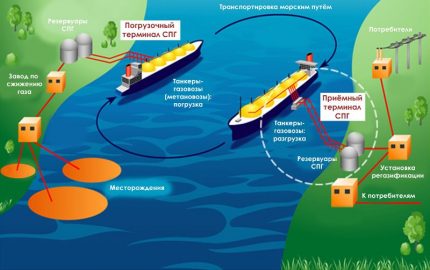
Most often, the terms “liquefied gas” and “liquefied gas” are mentioned in the context of the transportation of hydrocarbon energy. That is, the first production of blue fuel occurs, and then its conversion to LPG or LNG. Next, the resulting liquid is transported and then again returned to the gaseous state for a particular application.

LPG from propane-butane is mainly used as:
- gas engine fuel;
- fuel for injection into gas tanks of autonomous heating systems;
- liquids for refueling lighters and gas cylinders with a capacity of 200 ml to 50 liters.
LNG is usually produced exclusively for long-distance transport. If there is enough capacity to store LPG that can withstand pressure of several atmospheres, then for liquefied methane special cryogenic tanks are required.
LNG storage equipment is highly technological and takes up a lot of space. Using such fuel in cars is not profitable because of the high cost of the cylinders. LNG trucks in the form of single experimental models already drive on the roads, but in the passenger car segment this “liquid” fuel is unlikely to find wide application in the near future.
Liquefied methane as a fuel is now increasingly used in operation:
- railway locomotives;
- sea vessels;
- river transport.
In addition to being used as an energy carrier, LPG and LNG are also used directly in liquid form at gas and petrochemical plants. They make various plastics and other hydrocarbon-based materials.
Technologies for producing LPG and LNG
To convert methane from gas to liquid, it must be cooled to -163 ° C. But propane-butane liquefies already at -40 °C. Accordingly, technology and costs are very different in both cases.
The following technologies from different companies are used to liquefy natural gas:
- AP-SMR (AP-X, AP-C3MR);
- Optimized Cascade
- DMR;
- PRICO;
- MFC
- GTL et al.
All of them are based on the processes of compression and / or heat exchange. The liquefaction operation takes place at the plant in several stages, during which the gas is gradually compressed and cooled to the temperature of the transition to the liquid phase.
Gas mixture preparation
Before starting to liquefy raw natural gas, it is required to remove water, helium, hydrogen, nitrogen, sulfur compounds and other impurities from it. For this, the adsorption technology of deep purification of a gas mixture is usually applied by passing it through molecular sieves.
Then the second stage of preparation of the feedstock occurs, during which heavy hydrocarbons are removed. As a result, only ethane and methane (or propane and butane) with less than 5% impurities remain in the gas, so that this fraction can begin to be cooled and liquefied.
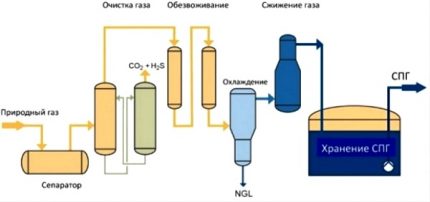
Fractionation allows you to get rid of harmful impurities and to allocate only the main gas for subsequent liquefaction. At a pressure of 1 atm, the transition temperature to the liquid state for methane is -163 ° С, for ethane -88 ° С, for propane -42 ° С, and for butane -0.5 ° С.
Just these temperature differences explain the reason why they are divided into fractions and only then they liquefy the gas entering the plant. There is no single liquefaction technology for all types of gaseous hydrocarbon compounds. For each of them it is necessary to build and apply their own production line.
The main process of liquefaction
The basis for converting gas to a liquid state is the refrigeration cycle, during which heat is transferred by one refrigerant or another from a medium with a low temperature to a medium with a higher one. This process is multistage and requires the availability of powerful compressors for expansion / contraction of the heat carrier and heat exchangers.
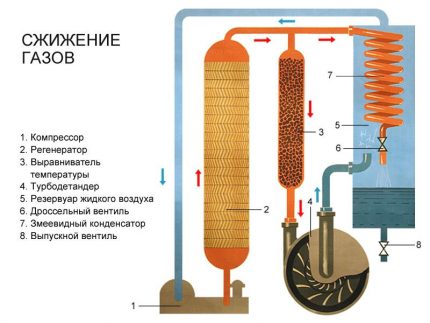
As a refrigerant at different stages of liquefaction, the following are used:
- propane;
- methane;
- ethane;
- nitrogen;
- water (sea and purified);
- air.
For example, for the primary cooling of natural gas at Yamal-LNG Novatek, cool Arctic air is used, which allows lowering the temperature of the feedstock with minimal costs immediately to +10 ° С. And in the hot summer months, instead of it, it is planned to use sea water from the Arctic Ocean, which, regardless of the time of year, is at a depth of 3-4 ° C.
At the same time, nitrogen obtained directly on the spot from the air is used as the final refrigerant on the Yamal Peninsula. As a result, the Arctic provides everything necessary for LNG production - from the source of natural gas to the working agents used in the liquefaction process.
Propane is liquefied in a similar manner to methane. Only cooling temperatures are required to it much less low - minus 42 ° C against minus 163 ° C. Therefore liquefaction gas for gas tanks costs several times less, but the resulting propane-butane LPG itself is less in demand on the market.
Transportation and storage
Almost the entire volume of LNG is transported by large sea gas tankers from one coast to another. Transportation by land is limited by the need to maintain the temperature of "liquid blue fuel" at values of about -160 ° C, otherwise methane begins to turn into a gas state and becomes explosive.

The pressure in the LNG tank is close to atmospheric. However, if the temperature of liquid methane rises above -160 ° C, then it will begin to turn from liquid to gas. As a result, the pressure in the tank will begin to rise, which poses a serious danger. Therefore, tankers for LNG transportation are equipped with installations for maintaining low temperatures and a powerful layer of heat insulator.
LPG is regasified into gas directly in the gas tank. And LNG regasification is carried out at special industrial plants without oxygen access. In physics, liquid methane is gradually converted to gas at a positive temperature. However, if this happens directly in the air outside of special conditions, then such a process will lead to an explosion.
After natural gas in the form of LNG is liquefied at the plant, it is transported, and then again at the plant (only regasification) is converted back into a gaseous state for further use.
Prospects for liquefied hydrogen
In addition to direct liquefaction and use in this form, one more energy carrier, hydrogen, can also be obtained from natural gas. Methane is CH4propane C3N8but butane C4N10.
The hydrogen component is present in all these fossil fuels, you just need to highlight it.
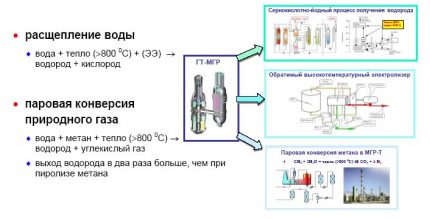
In order to transfer hydrogen from a gas state to a liquid, it needs to be cooled to -253 ° C. For this, multistage cooling systems and compression / expansion units are used. While such technologies are too expensive, but work is being done to reduce their cost.
We also recommend that you read our other article, where we described in detail how to make a hydrogen generator for your home with your own hands. More details - go link.
Also, unlike LPG and LNG, liquefied hydrogen is much more explosive. The smallest leak in connection with oxygen gives a gas-air mixture, which ignites from the slightest spark. And storage of liquid hydrogen is possible only in special cryogenic containers. There are still too many cons of hydrogen fuel.
Conclusions and useful video on the topic
How to produce liquefied gas and why it is liquefied:
All about liquefied gases:
There are several gas liquefaction technologies. They have their own for methane, and their own for propane-butane. At the same time, it is cheaper to get LPG, and transportation / storage is easier and safer. Obtaining methane LNG is a more expensive and complex process. Plus, its regasification requires specialized equipment. At the same time, methane is more in demand on the market today, so it is liquefied much in large volumes.
Do you have clarifying questions or your expert opinion on the topic of gas liquefaction? Perhaps you have something to add to the above. Feel free to ask and / or comment on the article in the box below.

 How much gas a gas stove consumes: gas flow calculation procedure
How much gas a gas stove consumes: gas flow calculation procedure  How much does it cost to connect gas to a private house: the price of organizing gas supply
How much does it cost to connect gas to a private house: the price of organizing gas supply  All about natural gas: composition and properties, production and use of natural gas
All about natural gas: composition and properties, production and use of natural gas  Gas consumption from the gas tank for heating: how to calculate + tips to minimize
Gas consumption from the gas tank for heating: how to calculate + tips to minimize  How to determine gas flow: methods for measuring and calculating the fuel used
How to determine gas flow: methods for measuring and calculating the fuel used  How and in what is gas flow measured: measurement methods + overview of all types of gas flow meters
How and in what is gas flow measured: measurement methods + overview of all types of gas flow meters  The best washing machines with dryer: model rating and customer tips
The best washing machines with dryer: model rating and customer tips  What is the color temperature of light and the nuances of choosing the temperature of the lamps to suit your needs
What is the color temperature of light and the nuances of choosing the temperature of the lamps to suit your needs  Replacement of a geyser in an apartment: replacement paperwork + basic norms and requirements
Replacement of a geyser in an apartment: replacement paperwork + basic norms and requirements